A) 0.28 M
B) 0.91 M
C) 1.1 M
D) 3.1 M
E) 3.9 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the percent CsCl by mass in a 0.711 M aqueous CsCl solution that has a density of 1.091g/mL?
A) 1.53%
B) 3.87 × 10-1%
C) 11.0%
D) 12.0%
E) 65.2%
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is defined as a solution that has a higher concentration of dissolved substances than plasma?
A) Hypotonic
B) Hypertonic
C) Aplasmic
D) Plasmic
E) Colloidal
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What name is given to the major component in a solution?
A) Supersaturated
B) Solute
C) Saturated
D) Unsaturated
E) Solvent
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mixing of solvent molecules and solute molecules is usually described as
A) the separation energy.
B) the heat of solvation.
C) an endothermic process.
D) an exothermic process.
E) the heat of solution.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is most soluble in CCl4?
A) NaCl
B) H2O
C) NaOH
D) C8H18
E) CH3OH
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Gases with high boiling points tend to be more soluble in water than ones with low boiling points.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the freezing point of a solution made from 22.0 g of octane (C8H18) dissolved in 148.0 g of benzene? (For benzene,freezing point = 5.50°C; Kf = 5.12°C/m)
A) -1.16°C
B) 0.98°C
C) 6.66°C
D) 12.2°C
E) 5.49°C
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molality of a 20.0% by mass ammonium sulfate (NH4) 2SO4 solution.The density of the solution is 1.117 g/mL.
A) 0.151 m
B) 0.200 m
C) 0.223 m
D) 1.51 m
E) 1.89 m
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What name is given to a minor component in a solution?
A) Solvent
B) Unsaturated
C) Saturated
D) Solute
E) Supersaturated
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substance is present in the largest proportion in a solution?
A) salute
B) gas
C) solvent
D) solid
E) solute
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is a strong electrolyte?
A) H2O
B) O2
C) H2SO4
D) C6H12O6 (glucose)
E) CH3COOH (acetic acid)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barbiturates are synthetic drugs used as sedatives and hypnotics.Barbital (184.2g/mol) is one of the simplest of these drugs.What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid? (For pure acetic acid,Kb = 3.07°C/m; boiling point of pure acetic acid = 117.9°C)
A) 117.0°C
B) 117.7°C
C) 118.1°C
D) 118.8°C
E) 120.4°C
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the value for the van't Hoff factor for all nonelectrolytes?
A) -1
B) 0
C) 1
D) ½
E) -½
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The distinguishing characteristic of all nonelectrolyte solutions is that they
A) contain ions.
B) do not conduct electricity.
C) react with other solutions.
D) always contain acids.
E) conducts heat.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The osmotic pressure of a 0.010 M MgSO4 solution at 25°C is 0.318 atm.Calculate i,the van't Hoff factor,for this MgSO4 solution. (R = 0.08206 L • atm/K • mol)
A) 0.013
B) 1.3
C) 1.5
D) 2.0
E) 76.8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances is the strongest electrolyte?
A) CO
B) CH3Cl
C) CH4
D) C2H5OH
E) HCl
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a diagram representing a solvent A(l) in a 1-L beaker,and a solute X dissolved in the solvent.Solvent A has a density of 0.8 g/mL,and a molar mass of 40 g/mol. Solute X has a molar mass of 30 g/mol.Each circle of X represents 1 mol of X.Assume that the solute addition does not significantly change the volume of liquid in the beaker. 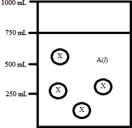 What is the mole fraction of the solute X in this solution?
What is the mole fraction of the solute X in this solution?
A) 0.67
B) 0.53
C) 0.27
D) 0.21
E) 0.04
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium nitrite is used as a corrosion inhibitor in lubricants.What is the molality of a solution prepared by dissolving 18.5 g of calcium nitrite in 83.5 g of distilled water?
A) 0.181 m
B) 0.222 m
C) 0.596 m
D) 1.35 m
E) 1.68 m
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to a solution that contains less solute than it has the capacity to dissolve?
A) Unsaturated
B) Saturated
C) Solvented
D) Oversaturated
E) Supersaturated
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 147
Related Exams