A) All of the carbon atoms in glucose came from carbon dioxide.
B) More atoms are present at the beginning than at the end.
C) More carbon dioxide is released from the plant than is absorbed.
D) Water is synthesized by the plant from H2 and O2.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four most common elements in living organisms are
A) C, H, O, Fe.
B) C, H, O, Na.
C) C, H, O, N.
D) C, N, O, Na.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A(n) ________ forms when two atoms share electrons.
A) ion
B) covalent bond
C) ionic bond
D) hydrogen bond
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You want to design an experiment to compare the effectiveness of Pepcid AC tablets and Alka-Seltzer tablets. Which of the following facts would be least likely to complicate your experiment and the analysis of your results?
A) PEPCID AC and Alka-Seltzer tablets contain different concentrations of the antacid drug.
B) PEPCID AC and Alka-Seltzer tablets contain different ingredients.
C) The recommended dosage for PEPCID AC and Alka-Seltzer tablets is different.
D) PEPCID AC and Alka-Seltzer tablets are sold in different countries throughout the world.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution with a pH of 7 is
A) strongly acidic.
B) weakly acidic.
C) neutral.
D) weakly basic.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Medicines are often administered in pill form. In many cases, the active ingredient of the pill (the drug) is joined to another substance by ________. This forms a(n) ________, which is stable in the dry environment of a pill bottle but dissociates under the wet conditions of the digestive system to release the drug to the body.
A) ionic bonds; salt
B) hydrogen bonds; base
C) ionic bonds; acid
D) covalent bonds; salt
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A nutrition facts label from a fortified cereal states that in a single serving of cereal there is 8% of the percent daily value of the element phosphorous. The label also says that a single serving of cereal plus one-half cup of skim milk has 20% of the percent daily value of phosphorus. Which of the following statements is false?
A) A half-cup of skim milk contains 12% of the percent daily value of phosphorus.
B) A half-cup of skim milk contains twice as much phosphorus as a serving of cereal.
C) Eating five servings of cereal and two and a half cups of skim milk will give you 100% of the phosphorus that you need for one day.
D) The amount of phosphorus in six servings of cereal is equal to the amount of phosphorus in two cups of skim milk.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following trace elements needed by humans is commonly added to table salt?
A) iodine
B) iron
C) magnesium
D) fluoride
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Silicon is an element that is found directly underneath carbon in the periodic table and thus has similar properties to carbon, including electron orbitals. How many electrons are in the valence shell of a silicon atom?
A) 1
B) 2
C) 4
D) 6
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Typically, nitrogen atoms are composed of electrons, protons, and neutrons. An isotope of nitrogen could
A) be positively charged.
B) be negatively charged.
C) have more protons than the usual nitrogen atom.
D) have more neutrons than the usual nitrogen atom.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
![Thin-layer chromatography is a method that can be used to separate molecules by their polarity. A mixture of molecules in a nonpolar liquid (the mobile phase) is added to the bottom of a piece of adsorbent material (the stationary phase) and allowed to migrate up the material. More-polar molecules interact more strongly with the stationary phase and thus migrate less. Less-polar molecules interact less strongly with the stationary phase and thus migrate more. Below are sample data for four different molecules from a thin-layer chromatography experiment. [Art: custom PowerPoint figure created by the author] -What can you conclude from these data? A) Molecule B contains many C-H bonds. B) Molecule C is the most polar molecule. C) Molecule D is more polar than molecule A. D) Molecule A is an ion.](https://d2lvgg3v3hfg70.cloudfront.net/TB6039/11eaa8fa_4abf_69ab_96ab_1fc7cb4d03d0_TB6039_00_TB6039_00_TB6039_00.jpg) Thin-layer chromatography is a method that can be used to separate molecules by their polarity. A mixture of molecules in a nonpolar liquid (the mobile phase) is added to the bottom of a piece of adsorbent material (the stationary phase) and allowed to migrate up the material. More-polar molecules interact more strongly with the stationary phase and thus migrate less. Less-polar molecules interact less strongly with the stationary phase and thus migrate more. Below are sample data for four different molecules from a thin-layer chromatography experiment. [Art: custom PowerPoint figure created by the author]
-What can you conclude from these data?
Thin-layer chromatography is a method that can be used to separate molecules by their polarity. A mixture of molecules in a nonpolar liquid (the mobile phase) is added to the bottom of a piece of adsorbent material (the stationary phase) and allowed to migrate up the material. More-polar molecules interact more strongly with the stationary phase and thus migrate less. Less-polar molecules interact less strongly with the stationary phase and thus migrate more. Below are sample data for four different molecules from a thin-layer chromatography experiment. [Art: custom PowerPoint figure created by the author]
-What can you conclude from these data?
A) Molecule B contains many C-H bonds.
B) Molecule C is the most polar molecule.
C) Molecule D is more polar than molecule A.
D) Molecule A is an ion.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a figure from an experiment that was performed to measure the effect of carbonate ion (CO32-) concentration on the calcification (growth) rate of coral reefs. At which point on the graph is the ocean pH the lowest? 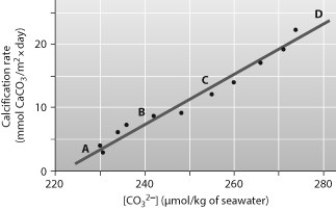
A) A
B) B
C) C
D) D
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As ice melts
A) hydrogen bonds are broken.
B) water molecules become less tightly packed.
C) the water becomes less dense.
D) heat is released.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrogen atom has one electron. How many covalent bonds can hydrogen form?
A) one
B) two
C) four
D) none
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following best describes the atomic number of an atom?
A) the number of protons in the atom
B) the number of electrons in the atom
C) the number of neutrons in the atom
D) the number of protons, electrons, and neutrons in the atom
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about water is false?
A) Ice is denser than liquid water.
B) Water naturally exists in all three physical states on Earth.
C) Floating ice on a pond insulates the liquid water below, slowing its rate of freezing.
D) If ice sank, the oceans would eventually freeze solid.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Uranium is a metallic element that is used in nuclear reactors and nuclear weapons. The vast majority of uranium found on Earth is in the form of uranium-238, an isotope with a mass number of 238, while the uranium that is used for nuclear reactors and weapons is uranium-235, an isotope with a mass number of 235. -What is the atomic difference between uranium-235 and uranium-238?
A) Uranium-235 has three fewer protons than uranium-238.
B) Uranium-235 has three fewer neutrons than uranium-238.
C) Uranium-235 has three fewer electrons than uranium-238.
D) Uranium-235 has three fewer atoms than uranium-238.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is dependent on the ability of water molecules to form hydrogen bonds with other molecules besides water?
A) the evaporative cooling of skin surfaces
B) the milder temperatures of coastal regions compared to inland areas
C) the ability of certain insects to walk on the surface of water
D) the universality of water as a solvent
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In ocean acidification, dissolving CO2 gas ________ the pH of the ocean.
A) lowers
B) does not affect
C) raises
D) triples
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about pH is true?
A) The pH scale is a measure of oxygen ion concentration.
B) A single unit change on the pH scale is equivalent to a 1% change in hydrogen ion concentration.
C) An increase in hydrogen ion concentration means a decrease in pH scale units.
D) Basic pH levels are less than 7.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 73
Related Exams