A) 3.44 eV
B) 1.51 eV
C) 3.02 eV
D) 0.75 eV
E) 4.53 eV
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy of a photon of visible light is on the order of
A) 10-6 eV
B) 10-3 eV
C) 1 eV
D) 103 eV
E) 106 eV
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
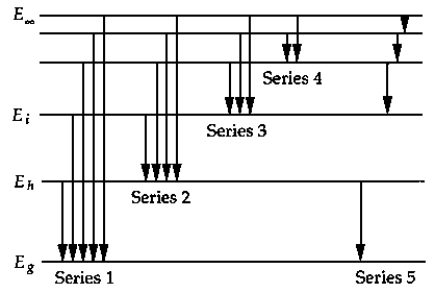 The above figure shows a schematic energy-level diagram for the hydrogen atom. The series that represents the Balmer series is
The above figure shows a schematic energy-level diagram for the hydrogen atom. The series that represents the Balmer series is
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A hydrogen atom is in a state with principal quantum number n = 3. A possible value for its orbital angular momentum is
A) zero
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of distinct electron states (including spin) when n = 5 is
A) 18
B) 22
C) 25
D) 50
E) 72
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the hydrogen atom in the ground state, the radial probability density is 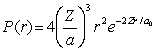 The probability of finding the electron in range r = 0.08a0 at r = 2a0 is approximately
The probability of finding the electron in range r = 0.08a0 at r = 2a0 is approximately
A) 2.34%
B) 3.67%
C) 5.86%
D) 6.25%
E) 7.43%
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The constant in the Rydberg-Ritz formula is RH = 10.96776 µm-1. The wavelength predicted by this formula for n1 = 3 and n2 = 2 is
A) 656 nm
B) 486 nm
C) 434 nm
D) 410 nm
E) None of these is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
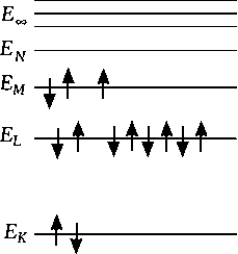 The above diagram shows the ionization energy and the shell distribution for an
The above diagram shows the ionization energy and the shell distribution for an  atom. To produce the K characteristic line, an incoming electron must have a minimum energy of
atom. To produce the K characteristic line, an incoming electron must have a minimum energy of
A) EL - EM
B) EK - EM
C) EK - EL
D) EN
E) EM
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first Bohr radius, r0, is 0.0529 nm and the corresponding energy, E0, is 13.6 eV. The wavelength of the light emitted as a hydrogen atom undergoes a transition from state n = 3 to n = 2 is
A) 656 nm
B) 486 nm
C) 434 nm
D) 410 nm
E) None of these is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
X rays are emitted when the electrons hit the screen in a cathode ray tube (CRT) , i.e., an old TV set. If the shortest wavelength X ray emitted is 3.7 *10-2 nm, then what is the voltage that the electrons are accelerated through in the TV set?
A) 2.1 * 10-23 V
B) 34 kV
C) 27 V
D) 34 V
E) 27 kV
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of electrons in the M shell for the element whose electronic configuration is 1s22s22p63s23p2 Must be
A) 2
B) 4
C) 6
D) 8
E) 10
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy of an X-ray photon of wavelength 3.0 * 10-10 m is
A) 1.0 *10-16 J
B) 9.9 * 10-16 J
C) 6.6 * 10-16 J
D) 2.2 *10-24 J
E) 3.2 * 10-29 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A K X-ray line of wavelength 0.1641 nm is emitted by a certain metal. What is the value of Z and which element is it?
A) 24, Cr
B) 25, Mn
C) 26, Fe
D) 27, Co
E) 28, Ni
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 The set of quantum numbers for the probability density shown is
The set of quantum numbers for the probability density shown is
A) n = 2, ![]() = 0, m = 0
= 0, m = 0
B) n = 2, ![]() = 1, m = 0
= 1, m = 0
C) n = 2, ![]() = 1, m = 1
= 1, m = 1
D) n = 2, ![]() = 0, m = 1
= 0, m = 1
E) n = 1, ![]() = 1, m = 0
= 1, m = 0
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to Bohr's model, the radius of an electron at n = 4 is
A) 0.212 nm
B) 0.846 nm
C) 0.0265 nm
D) 0.00331 nm
E) None of these is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The p state of an electronic configuration corresponds to
A) n = 2
B) l = 2
C) l = 1
D) n = 0
E) l = 0
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Bohr Model of the hydrogen atom, what is the ratio of the speed of the electron moving in the n = 2 orbit divided by the speed of the electron in the n = 3 orbit?
A) 0.66
B) 1.5
C) 2.25
D) 0.44
E) its speed is independent of n
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the angular momentum is characterized by the quantum number 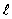 = 3, what are the possible values of Lz in units of
= 3, what are the possible values of Lz in units of 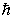 ?
?
A) -3, 0, +3
B) -3, -2, -1, 0
C) -2, -1, 0, +1, +2
D) -3, -2, -1, 0, +1, +2, +3
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy of the nth level in a one-electron atom is En = -13.6(Z2/n2) eV. Consider a beryllium ion with all but one of its electrons removed (a beryllium atom normally has four electrons) . What is the energy of the electron when it is in the third-lowest energy state?
A) -24 eV
B) -7.6 eV
C) -1.5 eV
D) 24 eV
E) 7.6 eV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The red line in the hydrogen emission spectrum is 656 nm. If the energy of the nth level is -13.6/n 2 eV, then calculate the transition between n levels that this emitted photon comes from.
A) n = 2 to n = 3
B) n = 4 to n = 3
C) n = 5 to n = 2
D) n = 3 to n = 2
E) n = 2 to n = 4
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 128
Related Exams