A) 1.30 * 10-5 g
B) 6.43 * 103 g
C) 7.80 * 1018 g
D) 1.56 * 10-4 g
E) 12.01 g
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 0.274 moles of a substance weighs 62.5 g, what is the molar mass of the substance, in units of g/mol?
A) 2.28 * 102 g/mol
B) 1.71 * 101 g/mol
C) 4.38 * 10-3 g/mol
D) 2.17 * 102 g/mol
E) none of these
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the percent composition by mass of sodium in Na2CO3.
Correct Answer

verified
Correct Answer
verified
Essay
Balance the following chemical equation: C4H10 + O2 CO2 + H2O
Correct Answer

verified
2C4H10 + 13 O...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Calculate the molecular mass of menthol, C10H20O.
A) 156 amu
B) 140 amu
C) 29 amu
D) 146 amu
E) 136 amu
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A chemistry student determined the empirical formula for titanium sulfide (TixSy).To do so, she reacted titanium with excess sulfur in a crucible, and recorded the following data: 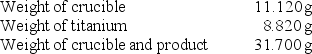 What is the empirical formula of titanium sulfide?
What is the empirical formula of titanium sulfide?
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the minimum mass of sulfur dioxide necessary to produce 200.g of sulfuric acid in the following reaction? 2SO2 + O2 + 2H2O 2H2SO4
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the theoretical yield of PI3 from the reaction of 27.0 g of P and 68.0 g of I2? 2P(s)+ 3I2(s) 2PI3(s)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sulfur atoms are there in 21.0 g of Al2S3?
A) 8.42 * 1022 atoms
B) 2.53 * 1023 atoms
C) 2.14 * 1023 atoms
D) 6.02 * 1023 atoms
E) 6.30 * 1026 atoms
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Phosphorus reacts with iodine as shown in the chemical reaction below.What is the percent yield of the reaction if 28.2 g PI3 is obtained from the reaction of 48.0 g of I2 with excess phosphorus? 2P(s)+ 3I2(s) 2PI3(s)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of Cl atoms are there in 65.2 g CHCl3?
A) 0.548 mol
B) 1.09 mol
C) 3.3 * 1023 mol
D) 1.64 mol
E) 3.0 mol
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One mole of iron
A) is heavier than one mole of lead (Pb) .
B) is 77.0 g of iron.
C) is 26.0 g of iron.
D) weighs the same as one mole of lead.
E) None of the above.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass of four moles of molecular bromine (Br2) is
A) 80 g.
B) 320 g.
C) 640 g.
D) 140 g.
E) 24 * 1023 g.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the average mass, in grams, of one potassium atom?
A) 5.14 * 10-23 g
B) 6.49 * 10-23 g
C) 6.02 * 10-18 g
D) 31.0 g
E) 39.1 g
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molecular formula of aspirin is C9H8O4. How many aspirin molecules are present in one 500-milligram tablet?
A) 2.77 molecules
B) 2.77 * 10-3 molecules
C) 1.67 * 1024 molecules
D) 1.67 * 1021 molecules
E) None of these is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of O atoms are in 25.7 g of CaSO4?
A) 0.189 mol
B) 0.755 mol
C) 4.00 mol
D) 1.14 * 1023 mol
E) 4.55 * 1023 mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
When a 0.952 g sample of an organic compound containing C, H, and O is burned completely in oxygen, 1.35 g of CO2 and 0.826 g of H2O are produced. What is the empirical formula of the compound?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following does not represent 1.00 mol of the indicated substance?
A) 6.02 * 1023 C atoms
B) 26.0 g Fe
C) 12.01 g C
D) 65.4 g Zn
E) 6.02* 1023 Fe atoms
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A method for producing pure copper metal involves the reaction of copper(I) sulfide with oxygen gas to give copper metal and sulfur dioxide. Suppose the yield of this reaction is 87%.What mass of a copper ore consisting of 46% copper(I) sulfide must be mined in order to produce 1.0 *103 kg (1.0 metric ton) of copper metal?
A) 1.4 * 103 kg
B) 3.2 * 103 kg
C) 1.3 * 103 kg
D) 1.5 * 103 kg
E) 8.0 * 103 kg
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Refer to the (unbalanced)equation CS2 + CaO CO2 + CaS. How many grams of CaS are produced if 53 g of CO2 are produced?
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 168
Related Exams