A) 84 K
B) 88 K
C) 90 K
D) 92 K
E) O2 doesn't boil because it is always a gas.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Which would have the stronger intermolecular forces of attraction HF or HCl?
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in NH3(l).
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in SO2(l).
Correct Answer

verified
dipole-dip...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which one of the following substances should exhibit hydrogen bonding in the liquid state?
A) SiH4
B) H2
C) H2S
D) CH4
E) CH3NH2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following in order of increasing boiling point: RbCl, CH3Cl, CH3OH, CH4.
A) CH3OH < CH3Cl < RbCl < CH4
B) CH3OH < CH4 < CH3Cl < RbCl
C) RbCl < CH3Cl < CH3OH < CH4
D) CH4 < CH3OH < CH3Cl < RbCl
E) CH4 < CH3Cl < CH3OH < RbCl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following characteristics indicates the presence of weak intermolecular forces in a liquid?
A) a low heat of vaporization
B) a high critical temperature
C) a low vapor pressure
D) a high boiling point
E) None of the above.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in HF(l).
Correct Answer

verified
hydrogen b...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The most space efficient arrangement of spheres is found in which type(s) of atom arrangement? I.hexagonal close-packed II.cubic close-packed III.simple cubic IV.body-centered cubic
A) I only
B) II only
C) I and II
D) IV only
E) I, II, and IV
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances crystallizes as a molecular solid?
A) KI
B) SiO2
C) Sn
D) CH3OH
E) Al2(SO4) 3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The shape of the water-to-glass meniscus results from the strong adhesive forces between glass and water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be expected to have the highest vapor pressure at room temperature?
A) ethanol, bp = 78°C
B) methanol, bp = 65°C
C) water, bp = 100°C
D) acetone, bp = 56°C
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The boiling points of chloromethane (CH3Cl) and dichlormethane (CH2Cl2) are - 24 °C and 40.°C respectively.The boiling point of trichloromethane (CHCl3) is predicted to be:
A) < - 24 °C
B) > 40.°C
C) > - 24 °C and < 40.°C
D) - 24 °C
E) 40.°C
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following data to determine the molar heat of vaporization of chlorine. 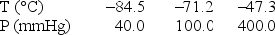
A) 34,700 J
B) 21,900 J
C) 9.99 kJ
D) 712 J
E) 317 J
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the temperature at which ethanol boils on a day in the mountains when the barometric pressure is 547 mmHg.(Given: The heat of vaporization of ethanol is 39.3 kJ/mol; the normal boiling point of ethanol is 78.3°C.)
A) 10.0°C
B) 69.9°C
C) 74.6°C
D) 76.5°C
E) 77.9°C
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
How much enthalpy is necessary to heat 10.0 g of solid benzene (C6H6)at 0.0°C to benzene vapor at 100°C? 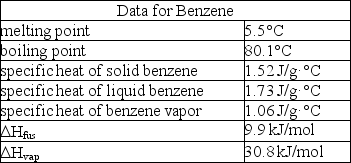
Correct Answer

verified
Correct Answer
verified
Essay
Polyethylene plastic consists of long chains of carbon atoms, each of which is also bonded to hydrogens as shown below: 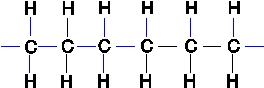 Water forms beads when placed on a polyethylene surface.Why?
Water forms beads when placed on a polyethylene surface.Why?
Correct Answer

verified
Water is highly polar and poly...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Indicate all the types of intermolecular forces of attraction in CF4(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The intermolecular forces present in CO include which of the following? I.dipole-dipole II.ion-dipole III.dispersion IV.hydrogen bonding
A) I, II, III, and IV
B) I and III
C) I, III, and IV
D) I and II
E) II and IV
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
MgO has the same crystal structure as NaCl, face-centered cubic.How many oxide ions surround each Mg2+ ion as nearest neighbors?
A) 4
B) 6
C) 8
D) 10
E) 12
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 149
Related Exams