A) closing container lid
B) increasing forces between molecules
C) increasing mass of molecule
D) decreasing temperature
E) decreasing surface area
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The relationship between the vapor pressure of a liquid and temperature can be expressed by the Clausius-Clapeyron equation: Ln[P2/P1] = [ΔvapH/R] × [1/T1 - 1/T2] Ethanol (C2H5OH) has a normal boiling point of 78.3 °C and,with ΔvapH = 39.3 kJ/mol.What is the vapor pressure of ethanol at 50.0 °C?
A) 118 Torr
B) 234 Torr
C) 354 Torr
D) 485 Torr
E) 670 Torr
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which probably has the lowest boiling point at 1.0 atm pressure?
A) PbH4
B) SnH4
C) SiH4
D) GeH4
E) CH4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following information,calculate ΔH° (in kcal mol-1) for: I(g) + e- → I-(g)

A) -633 kcal mol-1
B) -450 kcal mol-1
C) -71 kcal mol-1
D) -696 kcal mol-1
E) +712 kcal mol-1
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the following ionic compounds in order of increasing solubility in water: RbI,CaO,KCl
A) RbI,CaO,KCl
B) KCl,CaO,RbI
C) KCl,RbI,CaO
D) CaO,KCl,RbI
E) RbI,KCl,CaO
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Consider a parallelepiped with all edges being equal in length.There is an atom at each corner and one in the center.This is a simple cubic cell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the data below,determine the normal boiling point of liquid Rb. P1 = 400 mmHg,t1 = 620 °C,ΔvapH = 78.5 kJ/mol
A) 374 °C
B) 920 °C
C) 647 °C
D) 951 °C
E) 678 °C
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which combination of "type of solid" and specific example is INCORRECT?
A) ionic/"table salt"
B) metallic/copper wire
C) molecular/"dry ice"
D) network covalent/iodine
E) network covalent/silicon carbide
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The process in which a gas is transformed into a solid is called ________.
A) vaporization
B) condensation
C) solidification
D) deposition
E) fusion
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following forms an ionic solid?
A) Cu
B) C2H5NH2
C) NH4NO3
D) NO2
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Gold crystallizes in a face-centred cubic structure.What is the edge length of the unit cell if the atomic radius of gold is 144 pm?
A) 204 pm
B) 288 pm
C) 333 pm
D) 407 pm
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following describe the critical point of a liquid? I.the temperature and pressure at which a liquid's meniscus disappears II.the point where the vapor pressure curve intersects the fusion temperature curve III.the highest temperature and pressure at which a liquid can exist IV.the highest temperature at which it is possible to obtain a liquid from its vapor by increasing pressure
A) I,II,III
B) I,III,IV
C) II,III
D) II,III,IV
E) I,IV
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The maximum temperature at which a gas can be liquefied is just below the ________.
A) boiling temperature
B) critical temperature
C) melting temperature
D) normal temperature
E) superheating temperature
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements concerning molecules in the liquid state is true?
A) Cohesive forces are not important.
B) The molecules contract to fit the size of the container.
C) The molecules have no motion.
D) The molecules are in a patterned (oriented) arrangement.
E) The molecules are mobile and relatively close together.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of bonding does Sr form upon solidification?
A) covalent network
B) ionic
C) metallic
D) molecular
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The enthalpy of fusion is:
A) the quantity of heat required to melt one mole of a solid
B) the quantity of heat released when a solid melts
C) the quantity of heat when two elements are fused together to produce binary compound
D) the quantity of heat required to fuse two atomic nuclei
E) the quantity of heat released when an organic compound burns in an atmosphere of pure oxygen
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The heat of deposition equals the negative of the heat of sublimation.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many H- ions are around each Na+ ion in NaH,which has a cubic unit cell with H- ions on each corner and each face?
A) 1
B) 4
C) 6
D) 8
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound of iron and sulfur crystallizes in a lattice pattern described as cubic closest packed sulfide ions,with iron ions in all octahedral sites.What is its empirical formula?
A) FeS
B) FeS2
C) Fe3S4
D) Fe2S
E) Fe2S3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the phase diagram given,which of the following is INCORRECT? 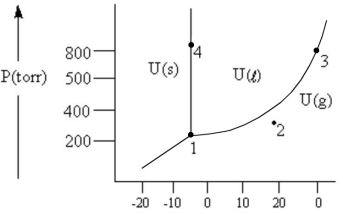
A) At the temperature and pressure of point 1,substance U exists as a three-phase equilibrium system.
B) At the temperature and pressure of point 2,substance U exists as a one-phase gaseous system.
C) At the temperature and pressure of point 3,substance U exists as a two-phase system.
D) If the U(s) ⇔ U(l) system is maintained at the temperature of point 4 while pressure is decreased steadily to about 300 Torr,more U will freeze.
E) There are no conditions of temperature and pressure under which solid U will vaporize without melting first.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 121
Related Exams