A) NaCl
B) NaF
C) CaBr2
D) CsI
E) CaCl2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following Lewis structure depicts the product when boron trifluoride combines with ammonia. How many total valence electrons are shown in this structure 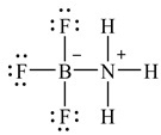
A) 28
B) 30
C) 32
D) 34
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in order of increasing ionic character: C - F O - F H - F Na - F
A) C - F < O - F < H - F < Na - F
B) C - F < H - F < O - F < Na - F
C) O - F < C - F < H - F < Na - F
D) H - F < C - F < O - F < Na - F
E) Na - F < H - F < C - F < O - F
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many covalent bonds will be drawn to phosphorous in PO43- for the dot structure that expands the octet to minimize formal charge and if necessary places negative formal charges on the most electronegative atom(s)
A) 4
B) 5
C) 6
D) 7
E) 8
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List all types of bonding present in the molecule SO3. I. ionic bond II. polar covalent bond III. nonpolar covalent bond
A) I only
B) II only
C) III only
D) I and II
E) II and III
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be a covalent compound
A) CsOH
B) NF3
C) Sr(NO3) 2
D) CaO
E) LiF
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The following Lewis dot symbol correctly depicts the chloride ion. 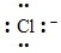
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in order of increasing ionic character:
A) B - C < C - N < C - O < C - C
B) C - C < B - C < C - N < C - O
C) C - C < C - N < B - C < C - O
D) C - O < C - N < B - C < C - C
E) C - N < C - C < B - C < C - O
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Each of the three resonance structures of NO3- has how many lone pairs of electrons
A) 7
B) 8
C) 9
D) 10
E) 13
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete this statement: Coulomb's law states that the magnitude of the between two charged bodies is
A) directly proportional to the product of the charges on the bodies and directly proportional to the distance separating them.
B) directly proportional to the product of the charges on the bodies, and inversely proportional to the square of the distance separating them.
C) inversely proportional to the product of the charges on the bodies, and directly proportional to the square of the distance separating them.
D) directly proportional to the sum of the charges on the bodies, and inversely proportional to the square of the distance separating them.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The following correctly depicts the Lewis dot symbol for the sulfide ion. 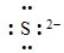
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of lone pairs in NCl3 is
A) 6.
B) 8.
C) 9.
D) 10.
E) 13.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has a Lewis structure that does not obey the octet rule
A) N2O
B) CS2
C) PH3
D) CCl4
E) NO2
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The molecule NH2NH2 shows both ionic and covalent bonding.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use bond energies to estimate the enthalpy change for the reaction of one mole of CH4 with chlorine gas to give CH3Cl and hydrogen chloride. BE(C-H) = 414 kJ/mol BE(C-Cl) = 326 kJ/mol BE(H-Cl) = 432 kJ/mol BE(Cl-Cl) = 243 kJ/mol
A) -106 kJ/mol
B) -101 kJ/mol
C) +101 kJ/mol
D) +106 kJ/mol
E) +331 kJ/mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound
A) NCl3
B) BaCl2
C) CO
D) SO2
E) SF4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following compounds does not follow the octet rule
A) NF3
B) CO2
C) CF4
D) Br2
E) NO
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of valence electrons in the molecule P2O5 is
A) 11.
B) 22.
C) 23.
D) 40.
E) 70.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis structure reveals a double bond in which of the following molecules
A) Br2
B) O2
C) N2
D) H2
E) CH4
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of resonance structures for the nitrate ion that satisfy the octet rule is
A) 1
B) 2
C) 3
D) 4
E) None of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 136
Related Exams