A) NaF < C3H8 < CH3CH2OH
B) C3H8 < NaF < CH3CH2OH
C) C3H8 < CH3CH2OH < NaF
D) CH3CH2OH < C3H8 < NaF
E) none of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ratio of the length of the body diagonal of a cube to the face diagonal of the cube?
A) 0.817
B) 1.00
C) 1.22
D) 1.41
E) 1.73
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
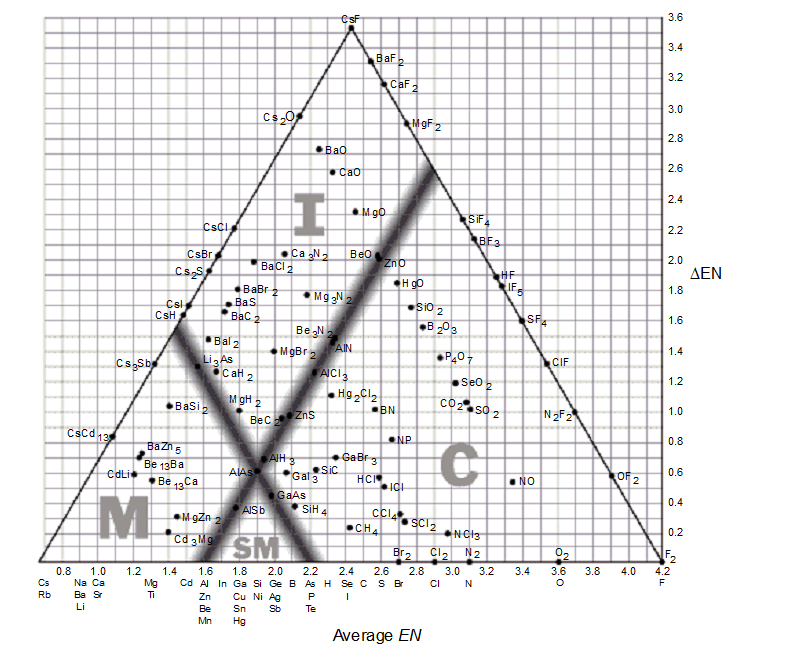 -Predict which compound has a high melting point and is a conductor in the molten and aqueous state.
-Predict which compound has a high melting point and is a conductor in the molten and aqueous state.
A) AlSb
B) AlN
C) AlLi
D) AlP
E) AlAs
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cesium iodide consists of a simple cubic lattice of l- ions with Cs+ ions in the center of the cubes. If the cell edge length is 0.445 nm, what is the Cs-I interatomic distance in nanometers?
A) 0.193 nm
B) 0.314 nm
C) 0.385 nm
D) 0.629 nm
E) 0.770 nm
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following types of solids represents the most ordered arrangement of repeating units?
A) polycrystalline
B) crystalline
C) hydrophobic
D) amorphous
E) none of these
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
 -Which of the following compounds will have the most metallic character?
-Which of the following compounds will have the most metallic character?
A) SnBr4
B) SnO2
C) SnP
D) SnS
E) SnCl4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate density in g/cm3 of titanium metal if the atomic radius of a titanium atom is 0.145 nm and the atoms form a body-centered cubic unit cell?
A) 0.1 g/cm3
B) 1 g/cm3
C) 5 g/cm3
D) 25 g/cm3
E) 30 g/cm3
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
A compound that is a poor conductor of electricity when solid, but a very good conductor when molten is most likely to fit into which category?
A) molecular solid
B) covalent solid
C) ionic solid
D) metallic solid
E) any of the above
G) None of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following types of solids is most likely to be soft, have a low melting point, and not conduct electricity when melted?
A) ionic
B) molecular
C) network covalent
D) metallic
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
 -Which of the following compounds will have the most covalent character?
-Which of the following compounds will have the most covalent character?
A) NaCl
B) SrCl2
C) PCl3
D) AlCl3
E) SnCl4
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following ionic compounds in order of increasing lattice energy (energy to break the solid into its gaseous ions) . MgF2, SrF2, BeF2
A) MgF2 < SrF2 < BeF2
B) SrF2 < MgF2 < BeF2
C) MgF2 < BeF2 < SrF2
D) SrF2 < BeF2 < MgF2
E) BeF2 < MgF2 < SrF2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
 -Predict which compound is an insulating ceramic material.
-Predict which compound is an insulating ceramic material.
A) MgZn2
B) Li3P
C) Li2O
D) LiCl
E) GaAs
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
 -Which of the following compounds will have the most ionic character?
-Which of the following compounds will have the most ionic character?
A) AlSb
B) AlN
C) AlLi
D) AlP
E) AlAs
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diamond (C(s) ) is a network covalent solid. Which of the following is most likely to be true of diamond?
A) Diamond would have a low melting point and would conduct electricity when melted.
B) Diamond would conduct electricity as a solid and its melting point cannot be predicted.
C) Diamond would have a high melting point and would conduct electricity when melted.
D) Diamond would have a high melting point and wouldn't conduct electricity when melted.
E) Diamond would have a low melting point and wouldn't conduct electricity when melted.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures has a coordination number of six?
A) simple cubic
B) body-centered cubic
C) cubic closest packed
D) hexagonal closest packed
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When diamond, NaCl, and water are boiled, the bonds being broken can best be described respectively as:
A) covalent, ionic, covalent
B) ionic, covalent, hydrogen bonds
C) covalent, ionic, hydrogen bonds
D) hydrogen bonds, ionic, covalent
E) none of the above
G) A) and E)
Correct Answer

verified
C
Correct Answer
verified
Short Answer
In KF, the K+ and F- ions are almost exactly the same size: 0.134 nm. Which would be larger: a neutral potassium atom or a neutral fluorine atom?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diamond is an example of:
A) a molecular solid
B) a network covalent solid
C) an ionic solid
D) a solid solution
E) a metallic solid
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the bond-type triangle for
 -Predict which compound is an electrical insulator and has a relatively low melting point.
-Predict which compound is an electrical insulator and has a relatively low melting point.
A) NaIn
B) SrCl2
C) PCl3
D) AlCl3
E) CaH2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which solid is held together by an extended network of covalent bonds?
A) sodium chloride
B) gold
C) calcium carbonate
D) diamond
E) dry ice (solid CO2)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 31
Related Exams