A) 18.75 %
B) 39.73 %
C) 68.69 %
D) 45.57 %
E) 34.44 %
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a quantitative analysis study, 4.624 grams of a hydrocarbon compound yielded 13.84 g of CO2 and 7.556 g of H2O in a combustion analysis apparatus. Determine the empirical formula of the compound. Hint: Use moles to relate the quantities of the substances.
A) CH3
B) C2H5
C) C3H8
D) C10H26
E) C10H27
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Ammonia reacts with diatomic oxygen to form nitric oxide and water vapor: 4NH3 + 5O2 → 4NO + 6H2O When 20.0 g NH3 and 50.0 g O2 are allowed to react, which is the limiting reagent?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic mass of cobalt is 58.933 u. How many moles of Co are there in a 7.60 g sample of cobalt?
A) 0.106 moles
B) 0.114 moles
C) 0.123 moles
D) 0.129 moles
E) 7.79 × 1022 moles
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct balanced chemical equation for the reaction below. C6H14 + O2 CO2 + H2O
A) 2C6H14 + 9O2 12CO2 + 7H2O
B) 2C6H14 + 19O2 12CO2 + 14H2O
C) 2C6H14 + 12O2 12CO2 + 14H2O
D) 3C6H14 + O2 18CO2 + 22H2O
E) C6H14 + O2 CO2 + H2O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
There are ________ moles of oxygen atoms in 25.7 g of CaSO4.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic mass of cobalt is 58.993 u. What is the mass of a cobalt sample that contains 0.763 moles of cobalt?
A) 39.7 g
B) 40.3 g
C) 43.4 g
D) 45.0 g
E) 45.7 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
In the course of determination of a chemical formula, a student obtained the following mole ratios: 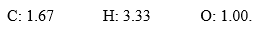 The empirical formula for the compound is ________.
The empirical formula for the compound is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the percent, by mass, of tungsten in W(CH3) 6?
A) 4.3819 %
B) 26.292 %
C) 67.083 %
D) 28.502 %
E) 35.722 %
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of carbon atoms are combined with 11.2 moles of hydrogen atoms in a sample of the compound, C3H8?
A) 3.00
B) 5.60
C) 4.20
D) 6.02 × 1023
E) 29.9
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 4.626 g sample of a hydrocarbon, upon combustion analysis, yielded 6.484 grams of carbon dioxide. What is the percent, by mass, of carbon in the hydrocarbon? Hint: Use moles to relate the quantities of the two substances.
A) 38.25 %
B) 19.47 %
C) 71.35 %
D) 40.16 %
E) 42.16 %
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound contains sodium, boron, and oxygen. An experimental analysis gave values of 53.976 % sodium and 8.461 % boron, by weight; the remainder of the mass is oxygen. What is the empirical formula of the compound? Hint: Assume you have 100 grams of this substance and use moles to relate the mass of one element to another.
A) NaBO2
B) Na3BO3
C) Na3BO2
D) NaB3O
E) Na3B3O8
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
An average atom of uranium (U)is approximately how many times heavier than an atom of phosphorus?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of carbon atoms are in 0.145 moles of the compound, C3H8?
A) 2.90 moles
B) 0.435 moles
C) 0.145 moles
D) 6.02 × 1023 moles
E) 29.9
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
In combustion analysis, a hydrocarbon compound undergoes complete combustion in excess oxygen to produce a stream of gases. The stream of gases passes through a pre-weighed tube. The increase in mass of these tubes represents the combined mass of CO2 and H2O. The mass of the element ________ in the hydrocarbon can be directly calculated from the mass of H2O.
Correct Answer

verified
Correct Answer
verified
True/False
In a chemical reaction, the best yield currently obtainable in any process is 99.5%.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
There are ________ moles of oxygen atoms in 10 moles of KClO3.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound contains arsenic and oxygen as its only elements. A 1.626 g sample of the compound contains 1.232 g of arsenic. What is the empirical formula of the compound?
A) As2O
B) AsO
C) As2O3
D) AsO2
E) As2O5
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
When the equation is balanced, Al2O3 + C → Al + CO2, the coefficient for CO2 is ________.
Correct Answer

verified
Correct Answer
verified
Short Answer
When octane (C8H18)is burned in a particular internal combustion engine, the yield of products (carbon dioxide and water)is 93%. What mass of carbon dioxide will be produced in this engine when 15.0 g of octane is burned with 15.0 g of oxygen gas?
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 207
Related Exams